
Institute for Protein Research, Osaka University, Japan. Protein structure is often described at four different scales. The secondary structure contains regions of amino acid chains that are stabilized by hydrogen bonds from the polypeptide backbone. The primary structure is comprised of a linear chain of amino acids. Only some proteins have a quaternary structure as well. Every amino acid also has another atom or group of atoms bonded to the. Every protein at least contains a primary, secondary, and tertiary structure. 
Each amino acid has the same core structure, which consists of a central carbon atom, also known as the alpha () carbon, bonded to an amino group (NH2), a carboxyl group (COOH), and a hydrogen atom. 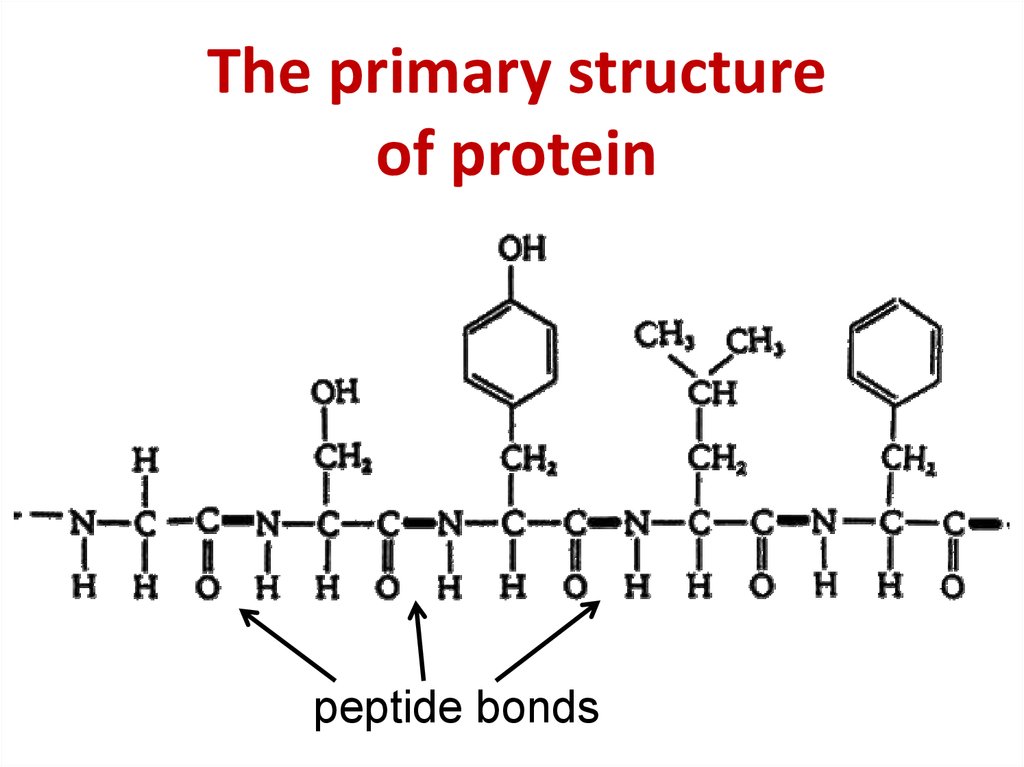
The presence of a disulfide bridge between the N-terminal extension Cys6 and Cys216 close to the putative active site in the C-terminal region is thought to be responsible for the generation of maximal proteolytic function in the pH range 8.5-10.7 and enhanced stability to denaturation. Amino acids are the monomers that make up proteins. It has also been shown that the protease has 9- and 26-residue extensions of the peptide chain at the N and C termini, respectively, and overall sequence homology is as low as 20% with bovine trypsin. Comparison of the amino acid sequence of Achromobacter protease and other serine proteases of bacterial and mammalian origins has revealed that Achromobacter protease I is a mammalian-type serine protease of which the catalytic triad comprises His57, Asp113, and Ser194. The protease consists of 268 residues with three disulfide bonds, which have been assigned to Cys6-Cys216, Cys12-Cys80, and Cys36-Cys58. This has been achieved by sequence analysis of the reduced and S-carboxymethylated protease and of peptides obtained by enzymatic digestion with Achromobacter protease I itself and Staphylococcus aureus V8 protease and by chemical cleavage with cyanogen bromide. The complete amino acid sequence of Achromobacter lyticus protease I (EC 3.4.21.50), which specifically hydrolyzes lysyl peptide bonds, has been established. Only a few structures existed then the only experimental method for protein structure determination was protein X-ray crystallography. Diversity, Equity, Inclusion, and Access For an extended period, the primary database for protein structures was the RSCB Protein Data Bank, created at the beginning of the 1970-ties.Citation, Usage, Privacy Policies, Logo.Biologically Interesting Molecule Reference Dictionary (BIRD).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
